An innovation for patients
with AADC deficiency
KEBILIDI is the FIRST AND ONLY gene therapy approved for patients with aromatic L-amino acid decarboxylase (AADC) deficiency
KEBILIDI is a gene therapy indicated for the treatment of adult and pediatric patients with aromatic L-amino acid decarboxylase (AADC) deficiency.
This indication is approved under accelerated approval based on a change from baseline in gross motor milestone achievement at 48 weeks post-treatment. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory clinical trial.

KEBILIDI is a one-time therapy that is delivered into the putamen, a part of the brain where the AADC enzyme is made
In AADC deficiency, a genetic variation causes a shortage in AADC enzyme production.
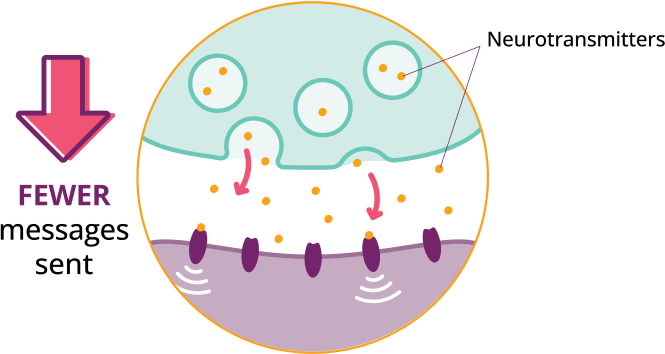
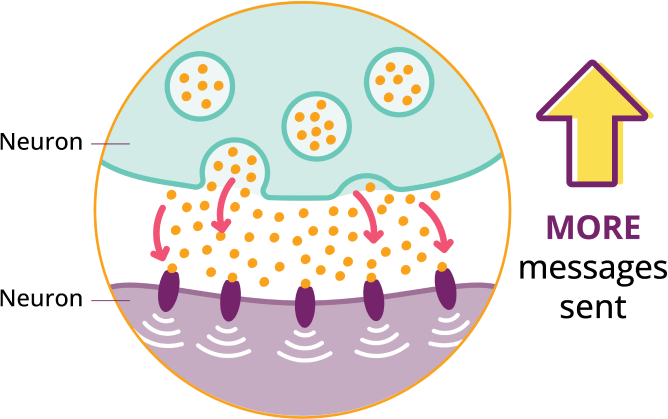
KEBILIDI delivers a working copy of the DDC gene to help cells produce the AADC enzyme and neurotransmitters your body needs, such as dopamine.
Are you interested in learning more about the services available through PTC Therapeutics? Visit PTCcares.com.
Locate a KEBILIDI Treatment Center
KEBILIDI is administered at designated treatment centers that specialize in pediatric neurosurgery.
Boston Children's Hospital
Boston, MA
Texas Children's Hospital
Houston, TX
Designated treatment centers are independently owned and operated. PTC Therapeutics does not have oversight of any designated treatment centers or the medical care they provide. Designated treatment centers decide whom they will evaluate for treatment based on various factors, including, but not limited to, the individual's age, insurance coverage, and capacity for new patients.
Inclusion of a designated treatment center in this locator tool does not represent an endorsement, referral, or recommendation from PTC Therapeutics.
No fees have or will be received by PTC Therapeutics for participation in the designated treatment center locator tool.
Visit PTCCares.com for more information.
Get Connected
Complete the form below, and a PTC Therapeutics team member will contact you.
Do you have a patient or loved one with AADC deficiency who may be eligible for KEBILIDI? Are you seeking more information? Would you like to receive updates about KEBILIDI? Would you like to speak to a member of the PTC Therapeutics team?
*Indicates required field.
Important Safety Information
What is KEBILIDI™ used for?
KEBILIDI is a gene therapy indicated for the treatment of adult and pediatric patients with aromatic L-amino acid decarboxylase (AADC) deficiency.
What Warnings should I know about KEBILIDI?
Procedural complications can occur with administration of KEBILIDI, which can include stopping of breathing or stopping of the heart within 24 hours of the procedure and during post-surgical care. Additional procedure related adverse events include cerebrospinal fluid leak, bleeding within the brain, inflammation of brain tissue, damage of brain tissue, and infection.
Uncontrolled movement was reported after administration of KEBILIDI. All events were reported within 3 months of administration and 2 events required hospitalization. Uncontrolled movements in the face, arm, leg, or entire body should be reported right away to a healthcare provider.
You should not use KEBILIDI if:
Skull maturity has not been achieved when assessed by neuroimaging, since skull maturity is needed for administration of KEBILIDI.
What are the side effects of KEBILIDI?
The most common side effects of KEBILIDI are uncontrolled movement, fever, low blood pressure, low red blood cell count, excess saliva production, low blood potassium, low blood phosphate, sleeplessness, low blood magnesium, and procedural complications, including stopping of breathing and stopping of the heart.
To report an adverse event, please call 1-866-562-4620 or email at usmedinfo@ptcbio.com. You may also report side effects to FDA at 1-800-FDA-1088 or at www.fda.gov/medwatch.
Please see for the full Prescribing Information for additional information.
